Clinical Trial Management Course
Clinical Trial Management Course - This course places a strong emphasis on the core principles of clinical trials management, covering essential areas such as site initiation, trial conduct, monitoring, and the effective. In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. You’ll learn about the computation of sample size and how to develop. Learn about regulatory requirements, data analysis, and patient safety. Refer to the data element definitions if submitting registration or results information. Gain an overview of principles and practices in clinical research from a global perspective. The aims of the course are: To develop attendees’ knowledge and skills in clinical trial management. In this course, you’ll learn more advanced operational skills that you and your team need to run a successful clinical trial. Explain the concept of clinical trial management (ctm) by understanding the fundamental concepts of study planning, study setup, conduct, and closeout. In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. Learn about regulatory requirements, data analysis, and patient safety. To develop attendees’ knowledge and skills in clinical trial management. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. Refer to the data element definitions if submitting registration or results information. The clinical trials management and regulatory compliance certificate provides rigorous clinical research training across the entire clinical trials process, from the perspective of the clinical. This course will provide you comprehensive training and develop skills needed to. Master clinical trial design and management for medical research. In this course, you’ll learn about the more advanced elements of managing clinical trials. Gain an overview of principles and practices in clinical research from a global perspective. The aims of the course are: In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. Master clinical trial design and management for medical research. This course will provide you comprehensive training and develop skills needed to. In this course, you’ll learn about. Gain an overview of principles and practices in clinical research from a global perspective. Master clinical trial design and management for medical research. This course places a strong emphasis on the core principles of clinical trials management, covering essential areas such as site initiation, trial conduct, monitoring, and the effective. From anticipating and planning for protocol events to conducting systematic. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. To improve attendees’ understanding of related disciplines including research. The aims of the course are: You’ll learn about the computation of sample size and how to develop. Explain the concept of clinical trial management (ctm) by understanding the fundamental concepts of study planning, study setup, conduct, and. Master clinical trial design and management for medical research. Learn about regulatory requirements, data analysis, and patient safety. This course places a strong emphasis on the core principles of clinical trials management, covering essential areas such as site initiation, trial conduct, monitoring, and the effective. In this course, you’ll learn about the more advanced elements of managing clinical trials. The. Learn about regulatory requirements, data analysis, and patient safety. To develop attendees’ knowledge and skills in clinical trial management. Enroll in sollers college's clinical trials management certification course. The clinical trials management and regulatory compliance certificate provides rigorous clinical research training across the entire clinical trials process, from the perspective of the clinical. From anticipating and planning for protocol events. Find your path at hmsonline coursestransform your careerleading harvard faculty In this course, you’ll learn more advanced operational skills that you and your team need to run a successful clinical trial. Learn about regulatory requirements, data analysis, and patient safety. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. In this course, you’ll learn about the. Enroll in sollers college's clinical trials management certification course. Learn about regulatory requirements, data analysis, and patient safety. Refer to the data element definitions if submitting registration or results information. In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. The clinical trials. This course places a strong emphasis on the core principles of clinical trials management, covering essential areas such as site initiation, trial conduct, monitoring, and the effective. To improve attendees’ understanding of related disciplines including research. This course will provide you comprehensive training and develop skills needed to. In this course, you’ll learn to collect and care for the data. The aims of the course are: To improve attendees’ understanding of related disciplines including research. To develop attendees’ knowledge and skills in clinical trial management. In this course, you’ll learn more advanced operational skills that you and your team need to run a successful clinical trial. Learn about regulatory requirements, data analysis, and patient safety. In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. Master clinical trial design and management for medical research. The clinical trials management and regulatory compliance certificate provides rigorous clinical research training across the entire clinical trials process, from the perspective of the. Gain an overview of principles and practices in clinical research from a global perspective. In this course, you’ll learn about the more advanced elements of managing clinical trials. You’ll learn about the computation of sample size and how to develop. Master clinical trial design and management for medical research. In this course, you’ll learn to collect and care for the data gathered during your trial and how to prevent mistakes and errors through quality assurance practices. This course will provide you comprehensive training and develop skills needed to. In this course, you’ll learn more advanced operational skills that you and your team need to run a successful clinical trial. To improve attendees’ understanding of related disciplines including research. Learn about regulatory requirements, data analysis, and patient safety. The aims of the course are: From anticipating and planning for protocol events to conducting systematic reviews to synthesize. Explain the concept of clinical trial management (ctm) by understanding the fundamental concepts of study planning, study setup, conduct, and closeout. Refer to the data element definitions if submitting registration or results information. Enroll in sollers college's clinical trials management certification course.Clinical Trial Management System (CTMS) PowerPoint and Google Slides
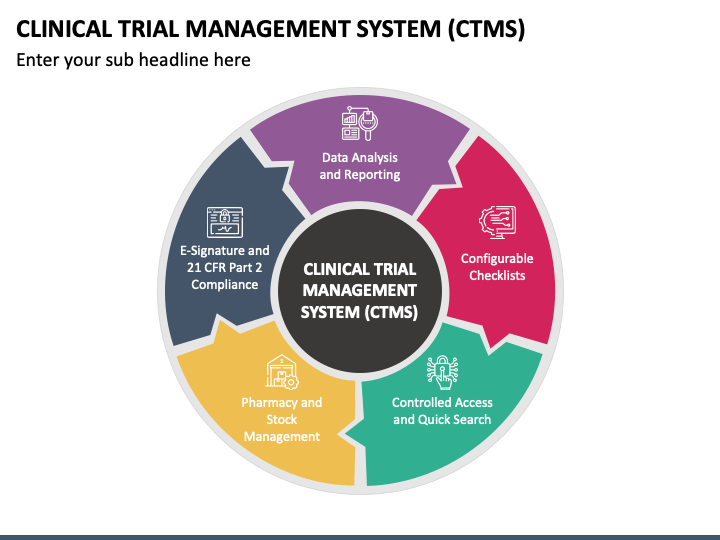
Clinical Trial Management System (CTMS) PowerPoint and Google Slides
PG diploma in Clinical Data Management Course Fees
What is a Clinical Trial Manager (CTM) Salary, Degree Requirements
Clinical Trial Management System Training Bangalore Clinical Research
Advance Your Career Path Clinical Trial Management Course
PPT Clinical Trial Management Services PowerPoint Presentation, free
ClinEssentials Clinical Trial Manager Training Course YouTube
Clinical trials management online course starts Feb. 19 University of
What is a Clinical Trial Management Software? Jelvix
The Clinical Trials Management And Regulatory Compliance Certificate Provides Rigorous Clinical Research Training Across The Entire Clinical Trials Process, From The Perspective Of The Clinical.
Find Your Path At Hmsonline Coursestransform Your Careerleading Harvard Faculty
This Course Places A Strong Emphasis On The Core Principles Of Clinical Trials Management, Covering Essential Areas Such As Site Initiation, Trial Conduct, Monitoring, And The Effective.
To Develop Attendees’ Knowledge And Skills In Clinical Trial Management.
Related Post: